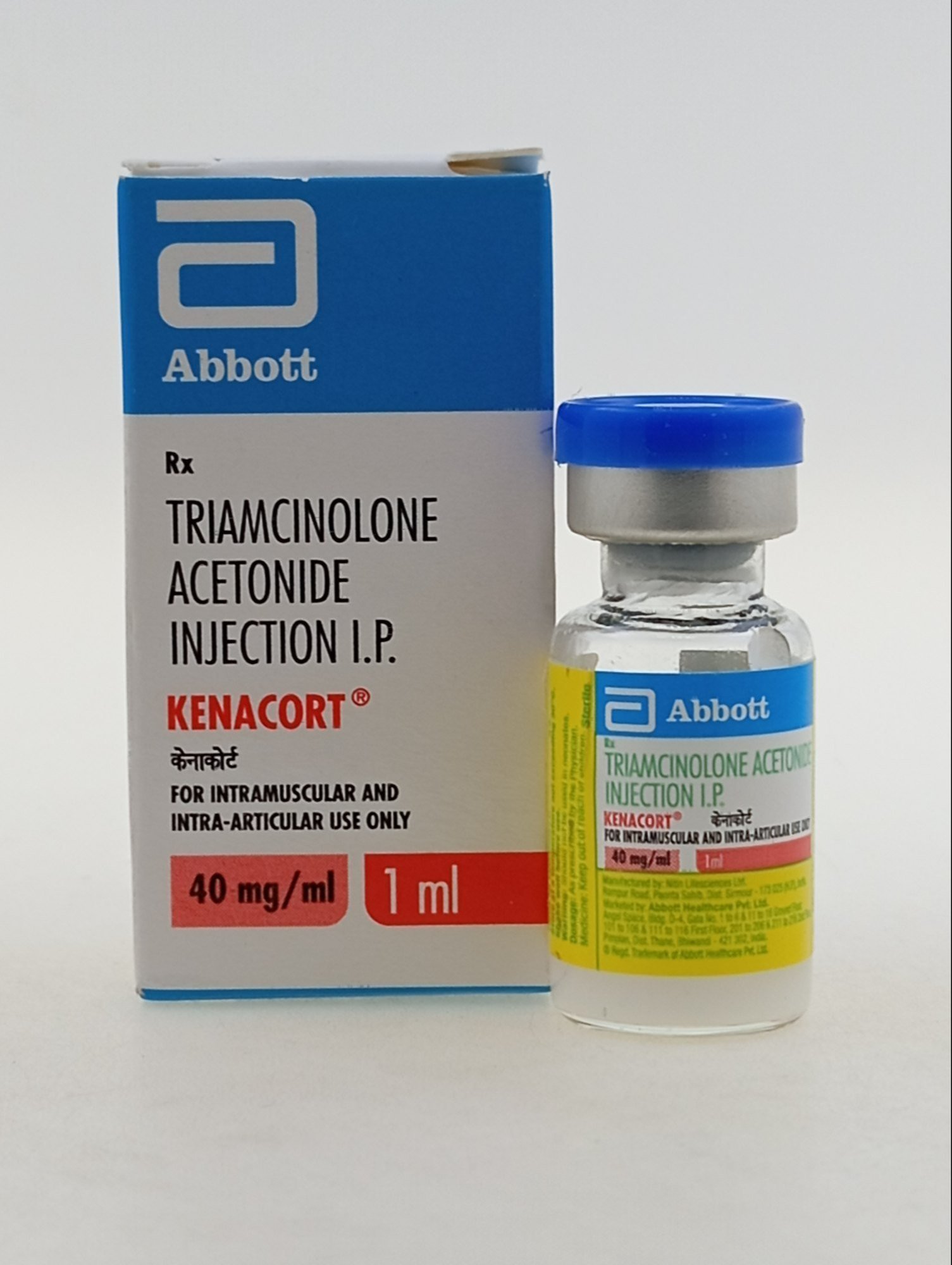
TRIAMCINOLONE ACETONIDE INJECTION
$22.00
| Feature | Technical Data |
|---|---|
| Active Ingredient | Triamcinolone Acetonide (USP) |
| Concentration | 40 mg per 1 mL |
| Vehicle | Sterile Aqueous Suspension (Sodium Chloride, Benzyl Alcohol) |
| pH Range | 5.0 to 7.5 |
| Potency Ratio | 5:1 compared to Hydrocortisone |
| Half-Life | 18–36 hours (Systemic); Weeks (Localized) |
| Classification | Glucocorticoid / Anti-inflammatory |
Triamcinolone Acetonide Injection is a potent, synthetic glucocorticoid designed to provide targeted and systemic relief for a diverse range of inflammatory disorders. Unlike short-acting oral steroids, this injectable suspension is engineered for extended-release, allowing the active ingredient to remain therapeutic within the body or a specific joint for several weeks.
Classified as a medium-to-long-acting corticosteroid, Triamcinolone Acetonide is roughly five times as potent as hydrocortisone but possesses significantly less mineralocorticoid activity. This means patients benefit from aggressive inflammation control with a reduced risk of the edema and salt retention often associated with earlier generations of steroid therapy. Whether used for intra-articular relief in chronic arthritis or intramuscularly for severe systemic allergies, it remains a cornerstone of modern clinical pharmacology.
Mechanism of Action: How it Works
At the molecular level, Triamcinolone Acetonide diffuses across cell membranes and complexes with specific cytoplasmic receptors. This complex then enters the cell nucleus, binds to DNA, and stimulates the transcription of mRNA. The result is the synthesis of enzymes—specifically lipocortins—which inhibit phospholipase A2.
By inhibiting this enzyme, the medication prevents the release of arachidonic acid, the precursor to potent inflammatory mediators like prostaglandins and leukotrienes.
-
Vascular Effect: It reduces capillary permeability and vasodilation.
-
Cellular Effect: It inhibits the migration of polymorphonuclear leukocytes and reverses increased capillary permeability.
-
Immune Effect: It suppresses the lymphatic system, reducing the production of antibodies and the immune response.
Primary Therapeutic Indications
1. Rheumatology and Orthopedics (Intra-articular)
For patients suffering from localized joint inflammation, Triamcinolone provides a “bridge” to recovery. By injecting directly into the synovial space, it provides rapid localized relief for:
-
Osteoarthritis: Reducing flare-ups when non-steroidal anti-inflammatory drugs (NSAIDs) are insufficient.
-
Rheumatoid Arthritis: Controlling acute localized inflammation in specific joints.
-
Bursitis & Synovitis: Rapidly reducing the swelling of the fluid-filled sacs surrounding the joints.
-
Epicondylitis (Tennis Elbow): Providing relief for chronic tendon-related pain.
2. Dermatology (Intralesional)
The suspension is highly effective when injected directly into skin lesions. It is the gold standard for:
-
Keloids and Hypertrophic Scars: Flattening scars and reducing itching.
-
Alopecia Areata: Stimulating hair regrowth in localized patches by suppressing the autoimmune attack on follicles.
-
Psoriasis: Targeting stubborn, localized plaques that resist topical creams.
-
Lichen Planus: Managing chronic inflammatory skin and mucosal conditions.
3. Systemic Conditions (Intramuscular)
When administered deep into the gluteal muscle, the medication is absorbed slowly into the bloodstream, providing weeks of coverage for:
-
Allergic States: Severe asthma, seasonal or perennial rhinitis (hay fever), and contact dermatitis.
-
Autoimmune Diseases: Systemic Lupus Erythematosus (SLE) and acute rheumatic carditis.
-
Endocrine Disorders: Primary or secondary adrenocortical insufficiency.
Detailed Product Specifications
| Feature | Technical Data |
|---|---|
| Active Ingredient | Triamcinolone Acetonide (USP) |
| Concentration | 40 mg per 1 mL |
| Vehicle | Sterile Aqueous Suspension (Sodium Chloride, Benzyl Alcohol) |
| pH Range | 5.0 to 7.5 |
| Potency Ratio | 5:1 compared to Hydrocortisone |
| Half-Life | 18–36 hours (Systemic); Weeks (Localized) |
| Classification | Glucocorticoid / Anti-inflammatory |
Administration Guidelines & Dosage
This product must be administered by a licensed healthcare professional.
-
Intramuscular (IM): The initial suggested dose for adults is 40 mg to 80 mg. It must be injected deep into the gluteal muscle to avoid subcutaneous atrophy (skin dimpling).
-
Intra-articular (IA): Depending on the size of the joint, doses range from 2.5 mg to 5 mg for small joints (fingers/toes) and 20 mg to 40 mg for large joints (knees/hips/shoulders).
-
Intralesional (IL): Usually limited to 1 mg to 3 mg per injection site, not to exceed 30 mg total in a single session.
Patient Safety & Contraindications
While highly effective, Triamcinolone Acetonide must be used with caution in certain populations:
-
Contraindications: Do not use in patients with systemic fungal infections or hypersensitivity to any component of the suspension.
-
Ocular Health: Use with caution in patients with ocular herpes simplex due to the risk of corneal perforation.
-
Cardiovascular: May cause elevation of blood pressure and salt/water retention in sensitive individuals.
-
Diabetes: Glucocorticoids can increase blood glucose levels; diabetic patients may require adjusted insulin dosages.
-
Pediatric Use: Long-term use can inhibit growth and development in children; use should be strictly monitored.
Storage and Handling
-
Temperature: Store at controlled room temperature between 20°C and 25°C.
-
Protection: Protect from light. Do not freeze, as freezing will cause the suspension to clump, making it impossible to inject safely.
-
Preparation: Shake the vial vigorously before use to ensure a uniform suspension. If the suspension has settled or hardened (caking), do not use.
The “ChemDope” Quality Assurance
Our Triamcinolone Acetonide is manufactured in GMP-certified facilities to ensure the highest standards of purity, sterility, and particle size consistency. We recognize that the efficacy of an injectable suspension depends heavily on the “micronization” of the active crystals—our formulation ensures smooth passage through fine-gauge needles and consistent absorption rates for predictable clinical outcomes.
Frequently Asked Questions (FAQ)
Q: How long does the pain relief last after a joint injection?A: Most patients begin to feel relief within 24 to 48 hours, with the peak effect occurring at the 1-week mark. Relief typically lasts between 4 to 12 weeks, depending on the severity of the condition and the patient’s activity level.
Q: Can this be used for “Bodybuilding” or performance?A: No. Triamcinolone is a corticosteroid (catabolic), not an anabolic steroid. It is used to reduce inflammation and actually breaks down muscle tissue if overused. It is strictly for medical therapeutic purposes.
Q: Is “Post-Injection Flare” normal?A: Some patients experience a temporary increase in joint pain for the first 24 hours after injection. This is often due to the crystal suspension irritating the joint lining before the anti-inflammatory effect takes hold. Cold compresses usually resolve this.